 
Antibodies to CTLA-4 may exert additional effects when used in vivo, by binding and thereby depleting regulatory T cells. Other groups who have analyzed the effect of antibodies to CTLA-4 in vivo have concluded little or no effect upon motility in the context of anergic T-cells. The disparity of these cells in multiple regards may explain some of these results. However, those studies compared CTLA-4 positive cells, which are predominantly regulatory cells and are at least partially activated, with CTLA-4 negative naive T cells. In this model CTLA-4 reverses the TCR-induced ‘stop signal’ needed for firm contact between T cells and antigen-presenting cells (APCs). ĬTLA-4 may also function via modulation of cell motility and/or signaling through PI3 kinase Early multiphoton microscopy studies observing T-cell motility in intact lymph nodes appeared to give evidence for the so-called ‘reverse-stop signaling model’. In addition to CTLA-4 CD80/CD86 interaction, fascin-dependent polarization of the cytoskeleton towards DC-Treg immune synapse may play a pivotal role. This suggests Treg-mediated immune suppression is a multi-step process. Although it is reversible upon T regulatory cell disengagement, this sequestration of essential cytoskeletal components causes a lethargic state of DCs, leading to reduced T cell priming. In addition to that, it has been found that dendritic cell (DC) - Treg interaction causes sequestration of Fascin-1, an actin-bundling protein essential for immunological synapse formation and skews Fascin-1–dependent actin polarization in antigen presenting DCs toward the Treg cell adhesion zone. More recent work has suggested that CTLA-4 may function in vivo by capturing and removing CD80 and CD86 from the membranes of antigen-presenting cells, thus making these unavailable for triggering of CD28. This work remains unconfirmed in the literature since its first publication. Biochemical evidence suggested that CTLA-4 recruits a phosphatase to the T cell receptor (TCR), thus attenuating the signal. The mechanism by which CTLA-4 acts in T cells remains somewhat controversial. 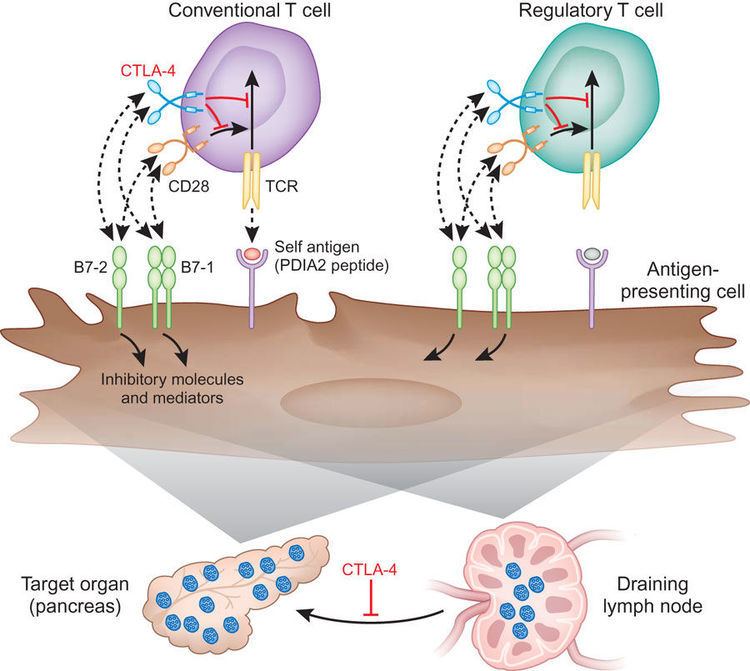
T cell activation through the T cell receptor and CD28 leads to increased expression of CTLA-4. CTLA-4 is also found in regulatory T cells (Tregs) and contributes to their inhibitory function. CTLA-4 transmits an inhibitory signal to T cells, whereas CD28 transmits a stimulatory signal. CTLA-4 binds CD80 and CD86 with greater affinity and avidity than CD28 thus enabling it to outcompete CD28 for its ligands. CTLA-4 is homologous to the T-cell co-stimulatory protein, CD28, and both molecules bind to CD80 and CD86, also called B7-1 and B7-2 respectively, on antigen-presenting cells. Function ĬTLA-4 is a member of the immunoglobulin superfamily that is expressed by activated T cells and transmits an inhibitory signal to T cells. Previous studies from several labs had used methods which could not definitively define the function of CTLA-4, and were contradictory. Sharpe independently published their findings on the discovery of the function of CTLA-4 as a negative regulator of T-cell activation, by knocking out the gene in mice. In November 1995, the labs of Tak Wah Mak and Arlene H. History ĬTLA-4 was first identified in 1991 as a second receptor for the T cell costimulation ligand B7. The CTLA-4 protein is encoded by the Ctla-4 gene in mice and the CTLA-4 gene in humans. It acts as an "off" switch when bound to CD80 or CD86 on the surface of antigen-presenting cells. CTLA-4 is constitutively expressed in regulatory T cells but only upregulated in conventional T cells after activation – a phenomenon which is particularly notable in cancers. ( September 2017) ( Learn how and when to remove this template message)ĬTLA-4 or CTLA4 ( cytotoxic T-lymphocyte-associated protein 4), also known as CD152 ( cluster of differentiation 152), is a protein receptor that functions as an immune checkpoint and downregulates immune responses. Please help improve it to make it understandable to non-experts, without removing the technical details. This article may be too technical for most readers to understand. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
